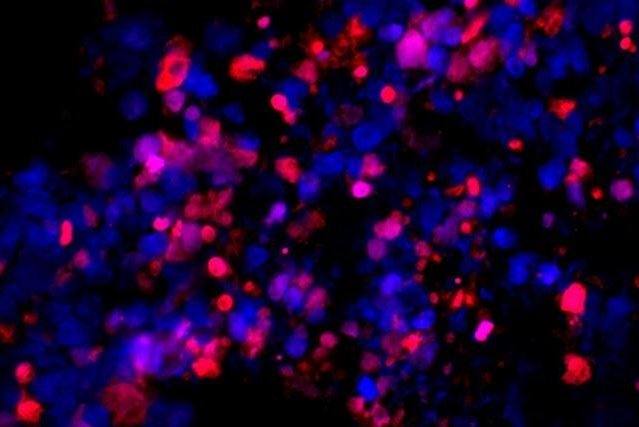
Metastatic human prostate cancer cells transplanted into a mouse self-destructed (red) when treated with an experimental drug that exposes cancer cells to their full, unhealthy appetite for protein synthesis. Image by
Ruggero lab/University of California San Francisco
May 3 (UPI) -- An experimental treatment was effective at causing treatment-resistant prostate cancer cells to self-destruct while leaving normal cells untouched, according to new research.
Researchers at the University of California San Francisco analyzed several hundred prostate tumors in mice and humans and found that the most aggressive cancers depend on a built-in cellular stress response. Their findings were published Wednesday in the journal Science Translational Medicine.
The researchers refer to their research as finding the "Achilles heel" for the resistant tumors.
"This is beautiful scientific work that could lead to urgently needed novel treatment strategies for men with very advanced prostate cancer," Dr. Peter Carroll, a UCSF Health prostate cancer surgeon, said in a press release.
About one man in nine will be diagnosed with prostrate cancer in his lifetime, with 1 in 41 dying from the disease, according to the American Cancer Society. Prostate cancer is the second-leading cause of cancer death for men in the United States behind lung cancer.
In the new study, the researchers found that prostate cancer's cellular stress responders can be stopped.
"We have learned that cancer cells become 'addicted' to protein synthesis to fuel their need for high-speed growth, but this dependence is also a liability: too much protein synthesis can become toxic," said senior study author Dr. Davide Ruggero, a professor of urology and cellular and molecular pharmacology at UCSF. "We have discovered the molecular restraints that let cancer cells keep their addiction under control and showed that if we remove these restraints they quickly burn out under the pressure of their own greed for protein."
Cancers often contain gene mutations that produce proteins at a high rate and cause self-destruction mechanisms, according to studies previously conducted by the UCSF researchers.
Because treatment-resistant prostate cancers often contain multiple mutations, they wanted to see how cancers can be contained from massive amounts of protein production.
To do so, they genetically engineered mice to develop prostate tumors containing two mutations seen in nearly 50 percent of patients with castration-resistant prostate cancer. The researchers discovered the highly aggressive cancers had lower rates of protein synthesis compared to milder cancers that have only a single mutation.
"I spent six months trying to understand if this was actually occurring, because it's not at all what we expected," said Dr. Crystal Conn, a postdoctoral researcher in the Ruggero lab.
The researchers report that they saw the same effect in mice, human cancer cell lines and 3-dimensional "organoid" models of the prostate.
They then found the combination of MYC and PTEN mutations trigger part of a cellular quality control system called the unfolded protein response. And they found these mutations alter the activity of a protein called eIF2a -- eukaryotic translation initiation factor 2a -- and turn it into an alternate form, P-eIF2a. This form slows down cellular protein production.
Researchers then examined 422 tumors surgically extracted from UCSF prostate cancer patients. They checked to see how these biomarkers predicted patient outcomes over 10 years of clinical follow-up. Only 4 percent of such tumors with low P-eIF2a spread after surgery, 19 percent of patients with high P-eIF2a went on to develop metastases, and many eventually died.
Researchers then looked to see whether blocking P-eIF2a's suppression of protein synthesis might effectively kill aggressive prostate cancers.
"Once we realized that these cancers are activating part of the UPR to put the brakes on their own protein synthesis, we began to ask what happens to the cancer if you remove the brakes," said Dr. Hao Nguyen, an assistant professor of urology at UCSF.
UCSF biochemist Peter Walter recently identified a molecule called ISRIB that reverses the effects of P-eIF2a activity. They tested it on on mice with prostate tumors and in human cancer cell lines.
The drug exposed aggressive cancer cells carrying combined PTEN/MYC mutations to an uncontrolled drive for protein synthesis, causing them to self-destruct. The drug didn't affect normal tissue, or even less-aggressive cancers lacking the MYC mutation.
Within three weeks, the prostate tumors began to shrink -- and didn't regrow after six weeks. Conversely, PTEN-only tumors had expanded by 40 percent.
The researchers implanted samples of human prostate cancer into mice, which has historically been unsuccessful in studies of prostate cancer. They found mice implanted with cells from the metastatic sample experienced dramatic tumor shrinkage and extended survival when treated with ISRIB. But mice implanted with cells from the less-aggressive primary prostate tumor saw the tumor only temporarily slow.
In addition, a third model was used to assess metastatic prostate cancer. It revealed that deaths from transplanted tumors, which kill mice within 10 days, were significantly reduced.
"This is very exciting because finding new treatments for castration-resistant prostate cancer is a pressing and unmet clinical need," Conn said.
The researchers are optimistic about clinical trials for ISRIB or related drugs that will attack advanced, aggressive prostate cancer.
"Most molecules that kill cancer also kill normal cells," Ruggero said. "But with ISRIB we've discovered a beautiful therapeutic window: normal cells are unaffected because they aren't using this aspect of the UPR to control their protein synthesis but aggressive cancer cells are toast without it."