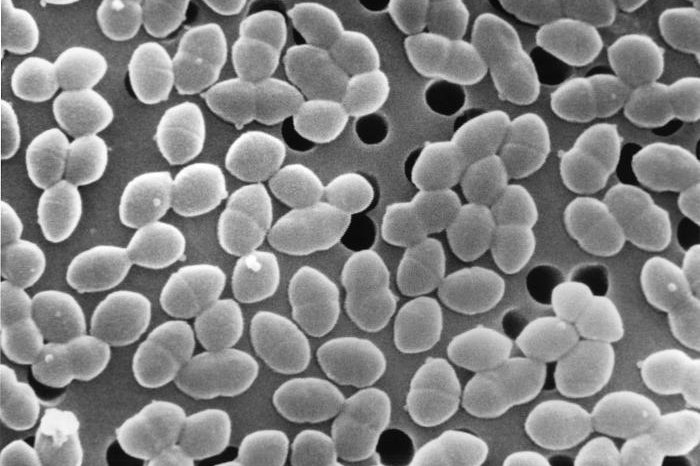
Enterococcus faecium, seen above, normally colonize the human digestive tract and gut but do not become pathogenic until a person's immune system has been compromised. The bacteria can become resistant to vancomycin if patients have been treated with either that or other antibiotics. Photo by Janice Haney Carr/CDC
NEW YORK, Feb. 25 (UPI) -- The overuse of antibiotics has given rise to a slowly increasing number of bacteria that cannot be controlled using the drugs. In patients who have been treated with antibiotics for an extended period of time, bacteria such as vancomycin-resistant enterococcus faecium, or VRE, can cause life-threatening infections.
Researchers at Sloan Kettering Medical Center have identified a molecule that was shown to reduce VRE in mice by mimicking the norovirus to enhance immune response in the body, according to a new study published in Science Translational Medicine.
The human body contains millions of bacteria, many of which are essential to basic function. Some bacteria can be harmful to humans, though some, such as VRE, often do not infect their hosts despite having colonized them. The immune system and other gut bacteria, which facilitate this colonization, generally prevent infection by VRE -- until either the immune system or gut microbiome is altered or compromised.
While the Centers for Disease Control and Prevention says VRE infections can often be fought using other antibiotics, patients who have been treated with types of antibiotics other than vancomycin, have been hospitalized, have weakened immune systems, and people who already have been colonized by the bacteria often do not respond to antibiotic treatment.
Based on previous studies, the researchers tested a murine norovirus in mice to boost antibacterial defenses in the gut and resist VRE colonization and infection. The norovirus induced an elevated expression of gene coding for Toll-like receptor 7, or TLR7, which is involved with antiviral and antibacterial immune pathways.
Needing to get around a concern that antibiotics, which help to cause VRE infection, would kill the norovirus before it did its job, the researchers identified a synthetic lignad called resiquimond, or R848, which mimics RNA in the virus that binds to TLR7. The researchers found that mice given R848 had significantly lowered VRE densities in their guts after being treated.
Researchers said more work is necessary because the long-term impacts of activating the TLR7 pathway are unknown, in addition to other fallout from changing gut microbiota.
"It's going to take a lot more research before it can get us to an actual therapeutic," Dr. Micahel Abt, a reseacher at Memorial Sloan Kettering Cancer Center, told The Scientist. "The encouraging thing is that, instead of using a whole commensal bacterium or a whole virus to stimulate the immune system, we're starting now to get to specific ligands that target specific pathways."