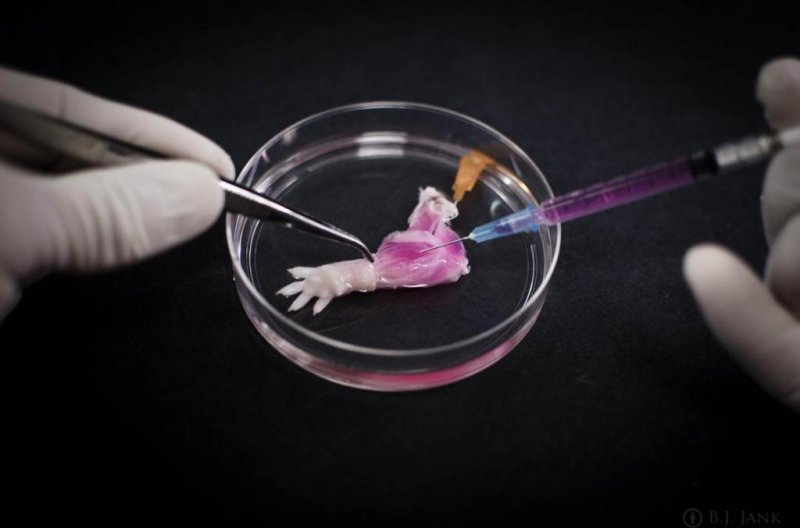
1 of 2 | A suspension of muscle progenitor cells is injected into the cell-free matrix of a decellularized rat limb, which provides shape and structure onto which regenerated tissue can grow. Photo by Bernhard Jank/Ott Laboratory, Massachusetts General Hospital Center for Regenerative Medicine
BOSTON, June 2 (UPI) -- Utilizing a method that has been used to successfully regenerate organs, researchers have regrown rat forelimbs and shown evidence that the same method could be used for the limbs of primates.
Researchers at Massachusetts General Hospital used limbs from deceased rats, stripping cells from the vascular and nerve matrix, and regrew muscles, veins and arteries. Electrical stimulation of muscle fibers caused them to contract with 80 percent of the strength a newborn animal would have, and blood flowed through the limb successfully when it was transplanted to a live animal.
"Limbs contain muscles, bone, cartilage, blood vessels, tendons, ligaments and nerves - each of which has to be rebuilt and requires a specific supporting structure called the matrix," Dr. Harald Ott of the MGH Department of Surgery and the Center for Regenerative Medicine, said in a press release. "We have shown that we can maintain the matrix of all of these tissues in their natural relationships to each other, that we can culture the entire construct over prolonged periods of time, and that we can repopulate the vascular system and musculature."
To turn the limb of a dead rat into one potentially transplantable into another living rat, the limb was put in a bioreactor and a detergent solution perfused through the vascular system to strip the limb of all cells. Once that process was complete, which took a week, researchers injected vascular cells into the main artery to begin regenerating arteries and veins while muscle progenitors were injected into matrix sheaths that define the position of each muscle.
After five days in the culture, electrical stimulation was used to promote growth. Two weeks later, the limb was removed from the bioreactor and tested, using electrical stimulation, and found to contract with 80 percent of the strength of a newborn animal. Once transplanted into a live animal, the veins and arteries filled with blood that circulated through the animal's body.
While a baboon forearm also was stripped of its cells using the same process as the rat limb, Ott said the next step will be to do a successful graft of a bioengineered limb and, taking note of successful hand transplants, see that nerves can also be regrown for full function.
"In clinical limb transplantation, nerves do grow back into the graft, enabling both motion and sensation, and we have learned that this process is largely guided by the nerve matrix within the graft. We hope in future work to show that the same will apply to bioartificial grafts," Ott said.
"Additional next steps will be replicating our success in muscle regeneration with human cells and expanding that to other tissue types, such as bone, cartilage and connective tissue."
The study is published in Biomaterials.